Polar or not polar? The interplay between reconstruction, Sr enrichment, and reduction at the La0.75Sr0.25MnO3(001) surface
Published:
Abstract
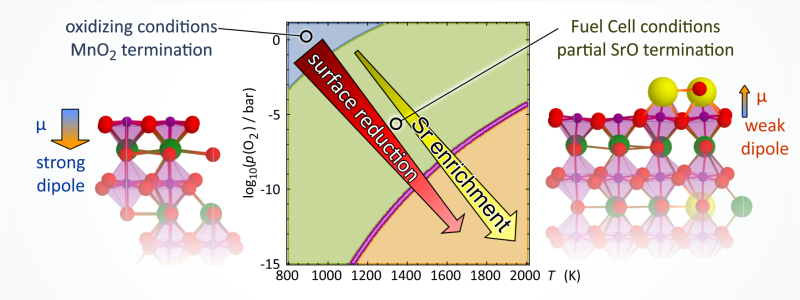
Perovskite oxides used in heterogeneous catalysis and electrocatalysis are tuned by substitutional doping. Sr-doped LaMnO3 is a popular choice of electrode material for electrochemical energy conversion. At elevated temperatures, relevant to solid oxide fuel or electrolysis cells, Sr enriches at the surface, which leads to fast degradation of the oxygen exchange activity at the surface. In this work, we investigate the effect of oxygen partial pressure p(O2) and temperature on Sr segregation at the La0.75Sr0.25MnO3(001) (LSM25) surface by ab initio thermodynamics calculations, taking into account different terminations and point defects Sr′′′La, V⋅⋅O, V′′′Mn, V′′′La, Oad, as well as associated defects of the type [V⋅⋅O−V′′′cation]. This model of the LSM25(001) surface addresses the connection between point defects and surface stability and makes quantitative predictions about the surface termination and Sr enrichment. Our results indicate that the MnO2 termination is stable under oxidizing conditions, while a partially covering SrO termination on MnO2 is stable for reaction conditions between 1000 and 1400 K and effective oxygen partial pressures between 10−11bar and 0.1 bar relevant to solid oxide fuel cell (SOFC) cathodes. Under more reducing conditions, we find that Sr is enriched at the surface regardless of surface termination. This trend is a direct consequence of the increased formation of positively charged oxygen vacancies at the surface, and this conclusion holds regardless of which surface termination is assumed. The partial SrO termination is exceptionally stable under SOFC cathode conditions because it minimizes the surface dipole density, while all the defect-free terminations of LSM25(001) are unstable due to surface polarity. This newly proposed termination leaves both Sr, as well as Mn and O sites of the MnO2 layer exposed at the surface and available for interaction with the gas phase. We propose that the LSM25(001) surface may consist of coexisting (La,Sr)O and MnO2 domains under SOFC cathode conditions, and we propose to include such a micropatterned surface in the future discussion of the active site in the oxygen reduction reaction.
Links
F. Hess and B. Yildiz. Phys. Rev. Mater. 4 (2020) 015801. 10.1103/PhysRevMaterials.4.015801
