Precipitation of dopants on acceptor-doped LaMnO3+-d revealed by defect chemistry from first principles
Published:
Abstract
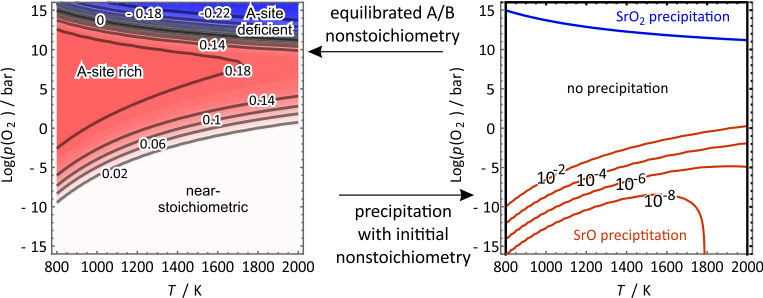
Perovskite oxides degrade at elevated temperatures while precipitating dopant-rich particles on the surface. A knowledge-based improvement of surface stability requires a fundamental and quantitative understanding of the dopant precipitation mechanism on these materials. We propose that dopant precipitation is a consequence of the variation of dopant solubility between calcination and operating conditions in solid oxide fuel cells (SOFCs) and electrolyzer cells (SOECs). To study dopant precipitation, we use 20% (D = Ca, Sr, Ba)-doped LaMnO3+δ (LDM20) as a model system. We employ a defect model taking input from density functional theory calculations. The defect model considers the equilibration of LDM20 with a reservoir consisting of dopant oxide (DO), peroxide (DO2), and O2 in the gas phase. The equilibrated non-stoichiometry of the A-site and B-site as a function of temperature, T, and oxygen partial pressure, p(O2), reveals three regimes for LDM20: A-site deficient (oxidizing conditions), A-site rich (atmospheric conditions), and near-stoichiometric (reducing conditions). Assuming an initial A/B non-stoichiometry, we compute the dopant precipitation boundaries in a p–T phase diagram. Our model predicts precipitation both under reducing (DO) and under highly oxidizing conditions (DO2). We found precipitation under anodic, SOEC conditions to be promoted by large dopant size, while under cathodic, SOFC conditions precipitation is promoted by initial A-site excess. The main driving forces for precipitation are oxygen uptake by the condensed phase under oxidizing conditions and oxygen release assisted by B-site vacancies under reducing conditions. Possible strategies for mitigating dopant precipitation under in electrolytic and fuel cell conditions are discussed.
Links
F. Hess and B. Yildiz. J. Chem. Phys. 154 (2021) 064702 . 10.1063/5.0035691
